Paxlovid, an antiviral therapy that is prescribed to COVID-19 patients to prevent severe illness, soon will become much more expensive to obtain.
As the medication transitions to the commercial market, Pfizer has set the updated price for a five-day course at $1,390 — more than double the $529 that the federal government had paid for the treatment.
The new pricing was announced in a letter Pfizer sent to pharmacies on Oct. 18, which was obtained by The Wall Street Journal.
COVID-19 REBOUND AFTER PAXLOVID TREATMENT LIKELY DUE TO INSUFFICIENT EXPOSURE TO THE DRUG
“Pricing for Paxlovid is based on the value it provides to patients, providers and health care systems due to its important role in helping reduce COVID-19-related hospitalizations and deaths,” Pfizer said in a statement.
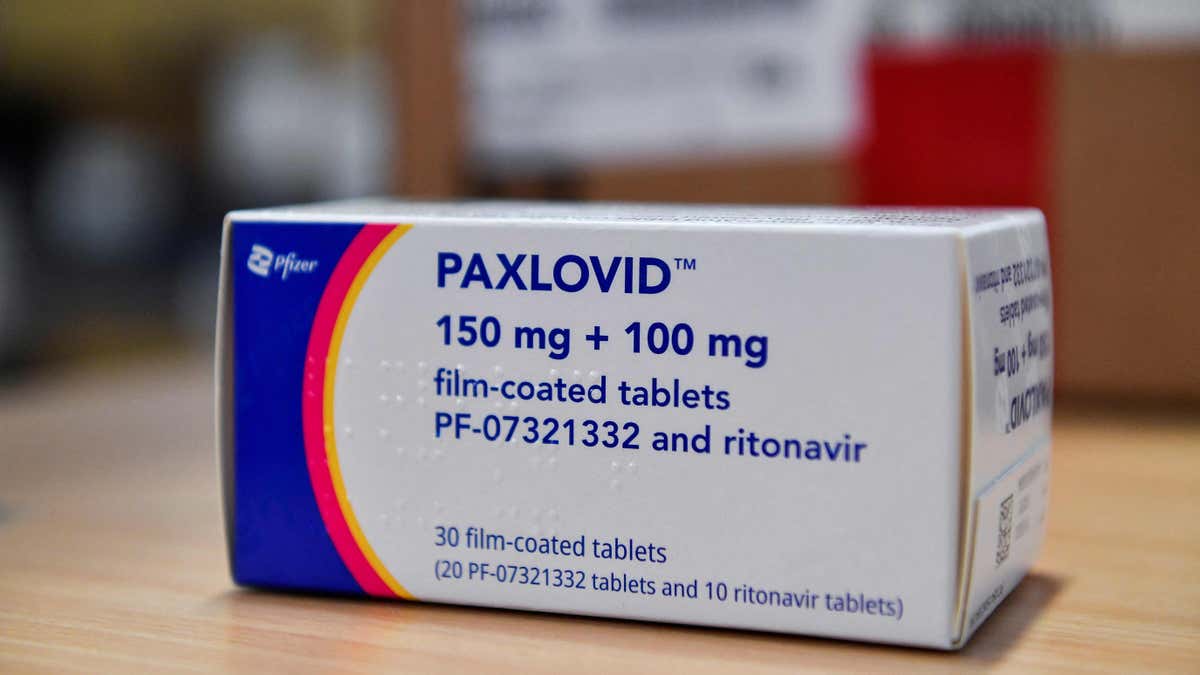
Last week, Pfizer announced in a release on its website that it “amended its supply agreement with the U.S. government for Paxlovid” as the emergency use authorization (EUA)-labeled medications are phased out.
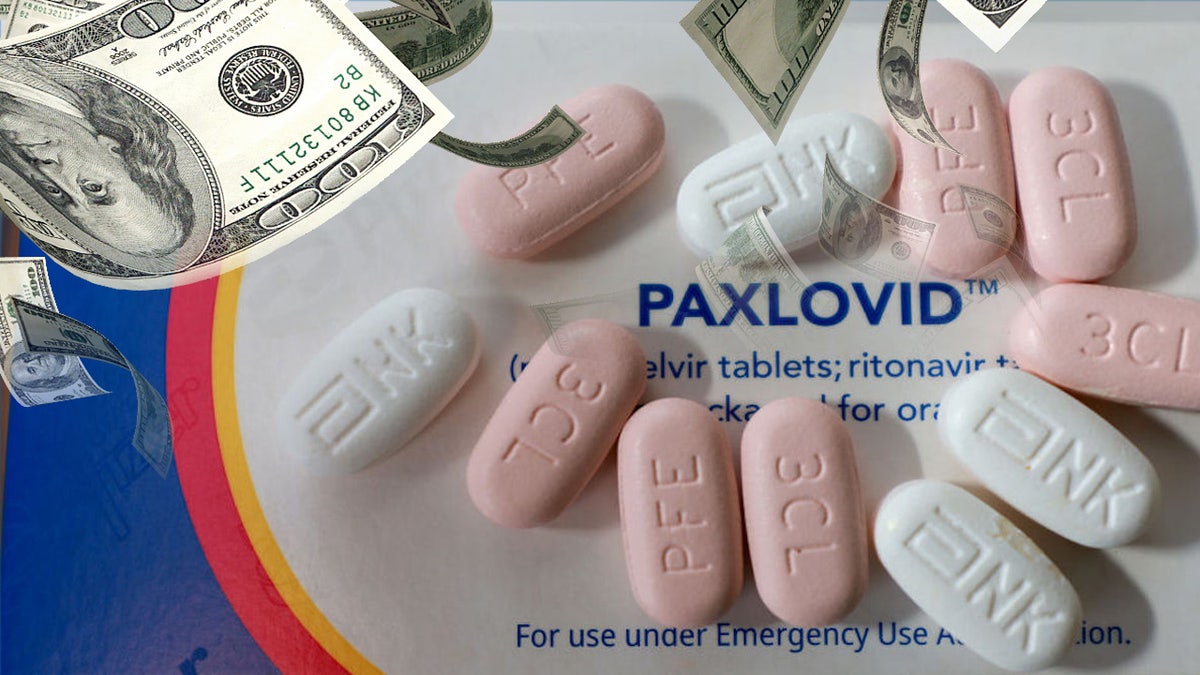
Paxlovid, an antiviral therapy that is prescribed to COVID-19 patients to prevent severe illness, soon will become much more expensive to obtain. (Getty Images)
Paxlovid’s transition to the commercial market — and to its updated pricing model — is set to begin in November of this year.
“The commercial transition will begin in November 2023, as the U.S. government begins to discontinue the distribution of EUA-labeled Paxlovid,” Pfizer wrote.
UPDATED COVID VACCINES: FDA ANNOUNCES EMERGENCY APPROVAL AND AUTHORIZATION
The company pointed out that the list price of the medication does not necessarily equate to what patients will pay out of pocket.
“As always, Pfizer’s goal is to ensure broad and equitable access to our medicines,” the company wrote in the letter.
“We are working diligently with payers to achieve the best possible formulary placement for Paxlovid, resulting in low out-of-pocket (OOP) costs for patients.”

Pfizer announced in a release on its website last week that it had “amended its supply agreement with the U.S. government for Paxlovid” as the emergency use authorization (EUA)-labeled medications are phased out. (iStock)
Pfizer said it also plans to offer a copay program for “eligible privately insured patients,” who could potentially get the drug for free.
In its own release, Pfizer also referenced a patient assistance program (PAP) in which patients on Medicare and Medicaid — as well as those who don’t have insurance — will receive Paxlovid free of charge through 2024.
CLICK HERE TO SIGN UP FOR OUR HEALTH NEWSLETTER
Paxlovid is a combination of two antiviral medications, nirmatrelvir and ritonavir.
Patients with mild to moderate cases of COVID take two doses of each drug for the course of the five-day treatment.
“The commercial transition will begin in November 2023, as the U.S. government begins to discontinue the distribution of EUA-labeled Paxlovid,” Pfizer wrote. (REUTERS/Jennifer Lorenzini/File Photo)
A statement from the U.S. Department of Health and Human Services (HHS) also announced Paxlovid’s upcoming transition.
“HHS will ensure a smooth and predictable transition to the commercial market over the next few months while prioritizing and reserving our HHS-procured treatment courses for people with Medicare and Medicaid, and for those who are uninsured,” the HHS wrote in the release.
CLICK HERE TO GET THE FOX NEWS APP
The date May 11, 2023, marked the official end of the federal public health emergency related to the COVID-19 pandemic.
However, the government’s declaration noted that emergency use authorizations for COVID-related products would not be affected.
“The timing to conclude the EUA is to be determined; it will not conclude on May 11, 2023, with the other declarations.”
Fox News Digital reached out to Pfizer for additional comment.
For more Health articles, visit www.foxnews.com/health.




More Stories
United Healthcare’s ransomware attack shows why supply chains are under siege
Nutrition Tips For Ramadan | JM Nutrition
Probiotics for IBS | The Nutritionist Reviews